
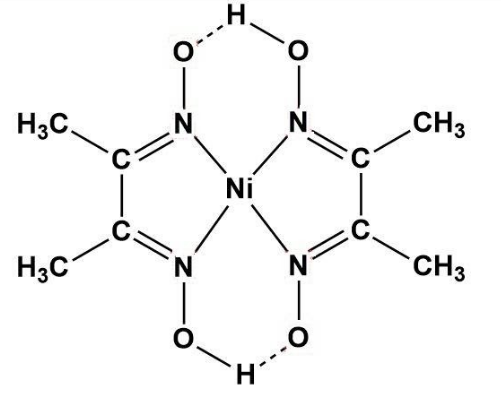
Figure 2 from Synthesis, physico-chemical and antimicrobial properties of Co(II), Ni(II) and Cu(II) mixed-ligand complexes of dimethylglyoxime - Part I | Semantic Scholar
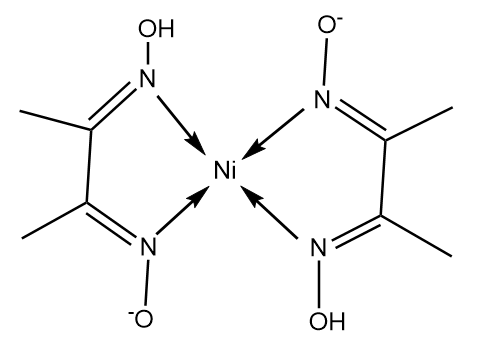
![Structure of dimethylglyoxime and nickel-dimethylglyoxime complex [179]. | Download Scientific Diagram Structure of dimethylglyoxime and nickel-dimethylglyoxime complex [179]. | Download Scientific Diagram](https://www.researchgate.net/publication/346253195/figure/fig4/AS:1022313969491973@1620750052886/Structure-of-dimethylglyoxime-and-nickel-dimethylglyoxime-complex-179.png)
Structure of dimethylglyoxime and nickel-dimethylglyoxime complex [179]. | Download Scientific Diagram


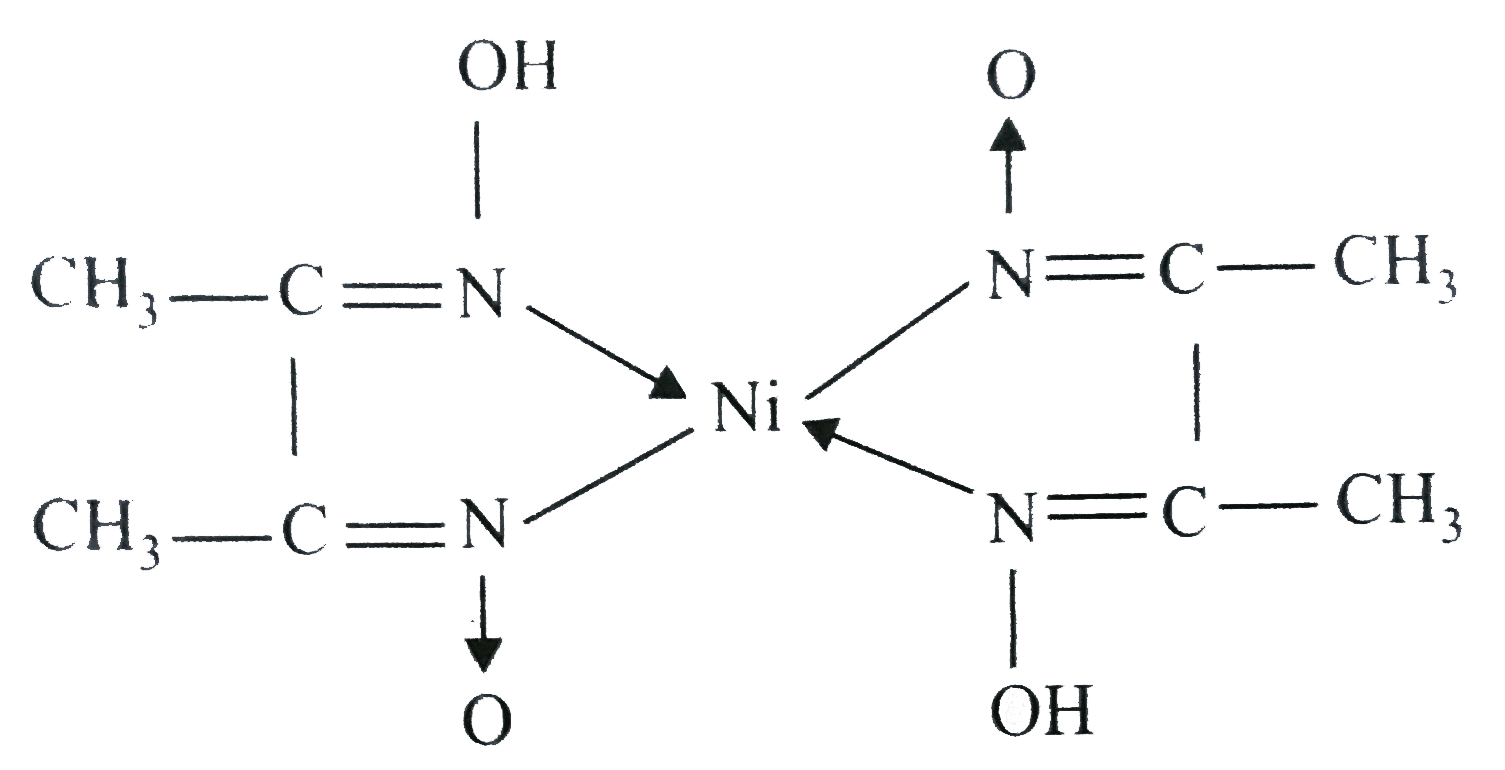


![What is the coordination number of Ni in the nickel-DMG complex?A.\\[2\\] B.$3$ C.$6$ D.$4$ What is the coordination number of Ni in the nickel-DMG complex?A.\\[2\\] B.$3$ C.$6$ D.$4$](https://www.vedantu.com/question-sets/ceab1c4b-aba0-43e1-b397-f3dbf0d0d9bd5238124797968177434.png)
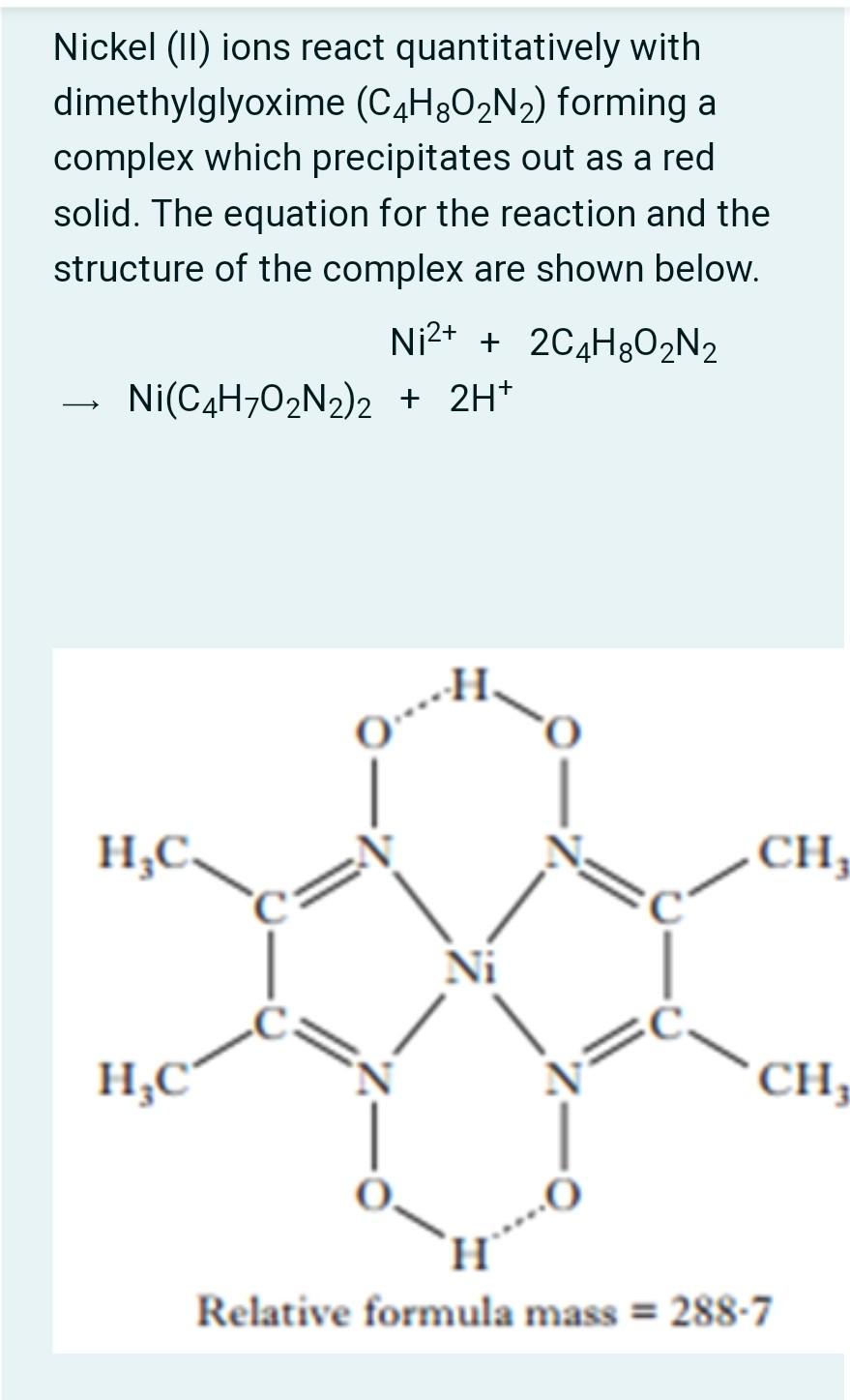
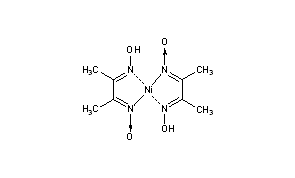





![The IUPAC name of the complex Ni[C(4)H(7)O(2)N(2)] formed by the react The IUPAC name of the complex Ni[C(4)H(7)O(2)N(2)] formed by the react](https://d10lpgp6xz60nq.cloudfront.net/physics_images/RS_P2_CHM_C09_E01_015_S01.png)
